
Original Article
Magnetic Drug Targeting Using Magnetic Nanoparticles for Cancer Therapy
|
1 Department of Biomedical Engineering, Engineering Faculty, Inonu University, Malatya, Turkey |
|
|
|
ABSTRACT |
||
|
Globally, cancer is still among the most frequent causes of mortality.Traditional cancer treatment modalities, including surgery, radiotherapy, and chemotherapy, present limited selectivity and severe side effects. Chemotherapeutic agents target the growth and survival of rapidly dividing cancer cells, but they can also kill healthy cells through systemic circulation. Hence, the quest for specific drug-targeting systems for tumor-targeting agents in the therapeutic arm has attracted considerable attention in cancer therapy. Magnetic nanoparticles (MNPs) have attracted significant attention in biomedical research owing to their specific physical and features such as their small particle size and high surface-to-volume, and superparamagnetic behavior. Magnetite (Fe₃O₄) and maghemite (γ-Fe₂O₃), which belong to the iron oxide nanoparticle family, are extensively investigated for biomedical because they are biocompatible and exhibit controllable magnetic properties. These nanoparticles can be functionalized with biocompatible coatings and anticancer agents to develop magnetic drug delivery systems. Magnetic Drug Targeting (MDT) is reliesed on the principle of magnetic nanoparticles conjugated with therapeutic agents directed to the cancerous tissue region by a magnetic field applied externall Such an approach makes increased drug accumulation within the targeted tissue possible, and a substantial impact has been achieved with systemic toxicity and side effects minimized. Furthermore, magnetic nanoparticles have been applied widely across biomedical fields, including magnetic resonance imaging (MRI), hyperthermia therapy, biosensors, and tissue engineering. The basic properties of magnetic nanoparticles, their biocompatibility, their application in cancer anticancer drug targeting using magnetic drug, and their properties in the context of the basic characteristics are investigated in this review as well. In addition, discussion has been provided on magnetic targeting dynamics, drug applications, and anti-cancer drugs with magnetic nanoparticles, as well as targeted drug system issues, based on the available literature. Drug delivery using magnetic nanoparticle systems is a promising strategy for optimizing cancer treatment while minimizing side effects. Keywords: Magnetic Drug Targeting, MDT, Cancer
Therapy, Magnetic Nanoparticles, MNPs, Targeted Drug Delivery |
||
INTRODUCTION
Conventional
cancer treatment methods mainly include surgical intervention, radiotherapy,
and chemotherapy. Among these treatments, chemotherapy—literally meaning
“treatment with drugs”—refers to the use of anticancer agents
(chemotherapeutics) that primarily target cancer cells Perry
(2008). In chemotherapy, both the type of drug and
its dosage are critical factors. The lack of selectivity and dose-dependent
toxicity of many anticancer drugs leads to significant challenges in
chemotherapy treatment. One of the most important problems is that anticancer
drugs destroy not only cancer cells but also all rapidly dividing cells in the
body through systemic circulation, including healthy cells Pardee
and Stein (2009). Furthermore, these treatments may lead to
organ-specific toxicity and severe side effects, such as heart failure in
breast cancer therapy Pugazhendhi
et al. (2018), Carvalho
et al. (2009), which may even result in life-threatening
conditions Alexiou
et al. (2011). The main goal of chemotherapy is to reduce
or eliminate the side effects of anticancer drugs while maximizing the drug
dose delivered to tumor tissue. Therefore, extensive research has focused on
drug-targeting systems that deliver lower drug doses to patients while
directing the drug specifically to cancerous tissue to achieve maximum
therapeutic effect Alexiou
et al. (2000). Magnetic drug targeting (MDT) systems using
magnetic nanoparticles represent an important approach to overcoming these
challenges in chemotherapy. In this strategy, chemotherapeutic drugs are
attached to iron-oxide cores and directed to cancerous tissue using a magnetic
field produced by an external source To prevent rapid elimination by the immune
defense system and to allow drug attachment, biocompatible materials must be
applied to the surface of magnetic nanoparticles Magnetic drug targeting (MDT)
systems using magnetic nanoparticles represent an important approach to
overcoming these challenges in chemotherapy. In this strategy, chemotherapeutic
drugs are attached to iron-oxide cores and directed to cancerous tissue using a
magnetic field produced by an external source To prevent rapid elimination by
the immune defense system and to allow drug attachment, surface modification of
magnetic nanoparticles with biocompatible materials is necessary.
Anticancer drugs
generally distribute widely throughout the body. Since anticancer drugs can
damage both tumor cells and healthy cells, the effectiveness of chemotherapy is
frequently restricted by severe side effects Brigger
et al. (2012). These side effects may range from
relatively temporary conditions such as nausea and hair loss to more severe
complications, including heart attacks. In some cases, treatment may even need
to be discontinued due to these adverse effects. Moreover, the success of
chemotherapy largely depends on the dosage of the administered drug.
Unfortunately, increasing the drug dose is not a complete solution, as higher
doses also lead to a significant increase in side effects. One of the major
concerns is how much of the anticancer drug administered into the systemic
circulation actually reaches the cancerous tissue. Drugs introduced into the
systemic circulation travel throughout the entire body, and only a minor
proportion of the therapeutic agent reaches the tumor region Consequently,
healthy tissues are also exposed to the effects of anticancer drugs. The
fundamental question that needs to be addressed is whether the administered
anticancer drug can be specifically directed to the cancerous tissue and
retained at that site. If this problem can be solved, much lower drug doses
could be used for treatment, and a larger proportion of the administered drug
could be delivered directly to the tumor region. At this point, the concept of
drug targeting, which involves the use of delivery vehicles to transport drugs
to particular sites in the body , becomes particularly important. In the early
1900s, the pioneer of immunology Paul Ehrlich introduced the concept of the
“magic bullet”, proposing the idea of selectively targeting diseased cells
without harming healthy tissues Perry
(2008) Canefe and
Duman (1994).
Magnetic Nanoparticles (MNP)
Across various
scientific and technological disciplines, nanoscale materials with uniform
morphology and narrow size distribution are widely required (Gubin, 2009; Gubin
et al., 2005). Among these nanoscale materials, magnetic nanoparticles (MNPs)
have gained substantial interest owing to their broad range of applications MNPs
typically have very small diameters, generally ranging from 1 to 100 nm, which
results in a surface-to-volume ratio. These nanoparticles can exhibit magnetic
behavior and be manipulated by an external magnetic field Among various types
of magnetic nanoparticles, magnetite (Fe₃O₄) exhibits
superparamagnetic properties and is therefore extensively applied in
applications such as biosensors, drug transport systems and magnetic
hyperthermia therapeutic approach. In contrast, maghemite
(γ-Fe₂O₃) is extensively used as a contrast agent in MRI
applications and as a biosensor surface coating owing to its high
biocompatibility Laurent
et al. (2008),Gupta and Gupta (2005). The types of magnetic nanoparticles and
their main properties are presented in Table 1
Table 1
|
Table 1 Magnetic nanoparticles and their main properties |
||
|
Nanomaterial |
Chemical Formula |
Key Property |
|
Magnetite |
Fe₃O₄ |
Superparamagnetic behavior |
|
Maghemite |
γ‑Fe₂O₃ |
High biocompatibility |
|
Cobalt ferrite |
CoFe₂O₄ |
High magnetic anisotropy |
|
Nickel ferrite |
NiFe₂O₄ |
Chemical stability |
|
Manganese ferrite |
MnFe₂O₄ |
Use as MRI contrast agent |
|
Iron nanoparticles |
Fe |
High magnetic moment |
Magnetic
nanoparticles derived from iron oxide, especially magnetite (Fe₃O₄)
and maghemite (γ-Fe₂O₃), are commonly employed in biomedical
fields owing to their unique physicochemical properties. Their nanoscale
particle size and extensive surface-to-volume ratio , and superparamagnetic
behavior provide remarkable advantages in a variety of biomedical fields. As a
result, these nanoparticles have attracted increasing attention, especially in
cancer diagnosis and therapeutic applications, and are considered promising
materials for both current and future biomedical technologies.
Magnetic Drug Carriers
Following the
development of the first magnetic polymer carriers in the 1970s, various
magnetic nanoparticle and microparticle carriers have been designed to deliver
drugs to specific sites in vivo. These carrier systems continue to be
optimized. In most cases, the magnetic part of the particle is covered with
biocompatible polymers including polyethylene glycol (PEG), polyvinyl alcohol
(PVA), and dextran in recent studies, inorganic coatings such as silica have
also been investigated the coating provides protection for the magnetic
particle against the surrounding environment and can also be modified with
molecules including carboxyl groups, biotin, avidin, carbodiimide, and other
functional molecules. They provide binding sites that enable cytotoxic drugs or
targeting antibodies to be attached to the carrier complex. Pankhurst
et al. (2003)
Carriers generally
display one of two structural configurations (i) a magnetic particle core,
usually magnetite (Fe₃O₄) or maghemite (γ-Fe₂O₃),
coated with a biocompatible polymer, or (ii) a porous biocompatible polymer
matrix within which magnetic nanoparticles are embedded. Recent developments in
carrier systems have largely focused on new polymeric or inorganic coating
layers applied to magnetite and maghemite nanoparticles Pankhurst
et al. (2009), Deng et al. (2003). Moreover noble metal coatings, particularly
gold, have also been examined. Alternative magnetic particles, including iron,
cobalt, and nickel nanoparticles, have also been explored in various studies,
as well as materials such as yttrium aluminum garnet Grasset
et al. (2001). Furthermore, cobalt/silica carriers have
been investigated for potential applications in ophthalmic surgery for retinal
repair Dahiya
and Dureja (2016), Pugazhendhi
et al. (2018).
The nanoscale size of magnetic nanoparticles (MNPs), generally ranging from a few to several tens of nanometers, represents one of their main advantages. This size range enables their use in studies involving biological structures such as cells with dimensions of about 10–100 μm, viruses with sizes generally between 20 and 450 nm, proteins (approximately 5–50 nm in size) , and genes (typically about 2 nm in width and 10–100 nm in length) Another important advantage of MNPs is their size-dependent superparamagnetic behavior. These nanoparticles are highly preferred because they can become magnetized when exposed to a magnetic field produced by an external source; however, they lose their magnetization once the external magnetic field is removed. Another advantage is that MNPs can be controlled by an external magnetic field, which enables numerous in vivo nano-bio applications, including magnetic drug targeting (MDT) Pankhurst et al. (2009), Bohara et al. (2016).
Biocompatibility
Magnetic carriers intended for biomedical applications should be water-based, biocompatible, non-toxic, and non-immunogenic Häfeli and Pauer (1999). The biocompatibility and potential toxicity of magnetic nanoparticles are largely influenced by their core composition and surface coatings. Iron oxide nanoparticles, particularly magnetite (Fe₃O₄) and its oxidized form maghemite (γ-Fe₂O₃), are the most widely utilized magnetic nanoparticles in biomedical applications. In contrast, strongly magnetic materials such as cobalt and nickel tend to be toxic and prone to oxidation, making them less appropriate for biomedical use. Tartaj et al. (2003)
The toxicity of the magnetic ions incorporated into the
core is a crucial aspect that must be considered in the design of magnetic
nanoparticles (MNPs). Surface
coatings provide two important protective functions: they inhibit the release
of toxic ions from the magnetic core and protect the core against oxidation and
corrosion. Among various nanomaterials,
iron oxide nanoparticles are the most frequently utilized for in vivo
biomedical applications, largely because iron is physiologically well tolerated
by the human body. This is partly due to the body's natural ability to process
excess iron and store it within the core of the iron-storage protein ferritin.
Other elements such as manganese (Mn) and zinc (Zn) are also important
components of the human body; however, their tolerable limits are significantly
lower than those of iron. Therefore, appropriate surface modification
strategies are required when these materials are used in nanoparticle systems.
After administration in vivo, MNPs are recognized by the body’s main defense
mechanism, the reticuloendothelial system (RES). This system plays an important
role in removing foreign substances from the bloodstream. During this process,
MNPs interact with plasma proteins produced by the reticuloendothelial system
(RES), which play a key role in the clearance of nanoparticles from the
bloodstream. Appropriate surface coatings can inhibit the adsorption of these
proteins, thereby extending the circulation time of MNPs and improving their
chances of reaching the target tissue.
Medical and Technological Applications of Magnetic Nanoparticles
Magnetic
nanoparticles (MNPs) have gained significant interest in the scientific
community because of their diverse applications They are particularly used in
biomedicine Pankhurst
et al. (2009), Pankhurst
et al. (2003), drinking water purification Simeonidis
et al. (2015), commercial applications Rai and Morris (2019), and magnetic storage media Sun et al. (2000), Ross (2001). In addition, MNPs are used in magnetic inks
for inkjet printing Voit et al. (2003), biosensing technologies Dave and Gao (2009), Perez et
al. (2002), and biosensor applications Rocha
(2014), Miller
et al. (2001). In
biomedical fields, magnetic nanoparticles are important carriers for drug
transport , drug delivery systems Jain et al. (2005) Chourpa
et al. (2005), and as MRI contrast agents Boutry e
t al. (2006), Chouly
et al. (1996). Furthermore, MNPs are widely used in
magnetic hyperthermia treatments Ortega
and Pankhurst (2013), Babincova
et al. (2008), controlled fabrication of biomaterials Gupta et
al. (2008), studies related to hearing Kuznetsov
et al. (2001) and vision Voltairas
et al. (2002), hippocampus research Schultheiss
et al. (1999), Voltairas
et al. (2002) gene therapy Scherer
et al. (2002), Plank et
al. (2003), magnetic twisting cytometry (MTC) Mijailovich
et al.(2002), tissue engineering Dobson
et al. (2002), Cartmell
et al. (2002), and magnetic cell separation techniques Zborowski
and Chalmers (2011).
Applications such
as magnetic cell separation Zborowski
and Chalmers (2011), Antfolk
and Laurell (2017), magnetic hyperthermia Ortega
and Pankhurst (2013), Schultheiss
et al. (1999), Perigo
et al. (2015), magnetic resonance imaging (MRI) Boutr et
al. (2006), Chouly
et al. (1996), and gene therapy Scherer
et al. (2002), Plank et
al. (2003) represent some of the biomedical fields in
which magnetic nanoparticles (MNPs) are widely utilized Figure 1.
Figure 1
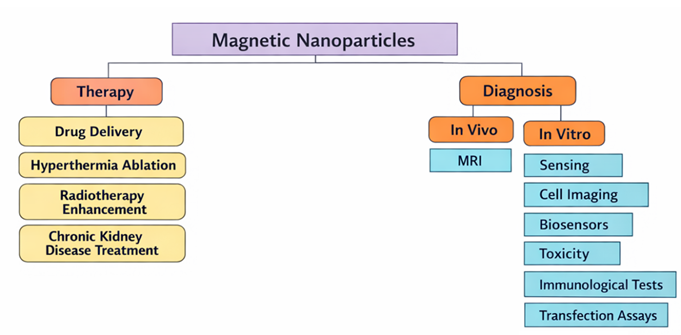
|
Figure 1 Role of Magnetic
Nanoparticles in Biomedical Applications (MNPs) |
Magnetic
nanoparticles (MNPs) used in biomedical applications must possess several
important properties, including strong magnetization, nanoscale particle sizes
(<100 nm), and a uniform particle size distribution, non-immunogenicity,
biocompatibility, water-based stability, and non-toxicity Laurent
et al. (2008), Häfeli
(2004).
Magnetic Drug Targeting
With the advancement of modern technology, nanotechnologies are now regarded as multimodal platforms capable of performing both diagnostic and therapeutic functions, commonly referred to as theranostics (Therapeutics + Diagnostics) Angelakeris (2017). Understanding the differences between normal cells and tumor cells is essential for the development of selective drug targeting strategies. The major reason for the failure in treating diseases like cancer is often associated with related not to drug discovery problems, but rather to drug delivery problems. Targeted delivery of the appropriate drug, at the correct place, time, and dosage, may greatly enhance treatment effectiveness for many diseases Since existing treatment options do not provide suitable solutions for every disease, advanced pharmacological technologies focus on developing patient-specific and targeted therapies designed to treat diseases effectively while minimizing toxicity Rai and Morris (2019). Advances in understanding the genetic alterations that transform normal cells into malignant cancer cells have led to the development of next-generation targeted therapies. By specifically targeting the biological abnormalities present in cancer cells, these therapies theoretically allow a greater number of normal cells to be spared from toxicity Pardee and Stein (2009).
Compared to bulk materials, nanomaterials possess distinct mechanical, electrical, optical, and magnetic properties. The utilization of these unique properties of nanomaterials helps address challenges related to the optimal location, timing, dosage, and delivery of therapeutic agents. Therapeutic approaches based on nanomaterials are designed to achieve improved treatment outcomes while requiring lower drug doses compared to conventional drug formulations Rai and Morris (2019).
In general, the main challenge in the cytotoxic approach commonly used in cancer therapy is how to minimize toxicity while maximizing anticancer activity Pardee and Stein (2009). To improve therapeutic outcomes without increasing toxicity, the drugs used should be relatively more specific to tumor cells than to normal tissues. Therefore, there is a growing need to develop new therapeutic strategies that may increase the efficiency of chemotherapy while limiting its side effect Developing strategies capable of selectively delivering drugs to diseased tissues while avoiding elevated concentrations in healthy tissues has become one of the most active areas in cancer research. Among these approaches, Magnetic Drug Targeting (MDT) systems have gained significant attention as a promising strategy for cancer management treatment. In MDT systems, anticancer drugs are attached to magnetic nanoparticles (MNPs) Alexiou et al. (2011), Lübbe et al. (1996), Alexiou et al. (2002). The fundamental principle of this approach is to administer a lower dose of the anticancer drug to the patient while directing it to tumor tissue subjected to an external magnetic field , thereby maximizing the therapeutic effect. Thus, the primary objective of MDT is to reduce or eliminate the side effects of anticancer drugs while delivering the maximum possible drug dose to tumor tissues. One of the greatest advantages of MDT is its potential to minimize chemotherapy-related side effects by directing smaller drug doses directly to the target cancer tissue without prolonged systemic circulation Bilgili (2022).
The application of magnetic nanoparticles (MNPs) in biomedicine was initially reported by Gilchrist in 1957, who administered maghemite particles into the lymph nodes surrounding surgically excised tumors, thereby enabling selective inductive heating of the lymph nodes Gilchrist et al. (1957). However, in this study, Gilchrist did not propose the magnetic guidance or targeting of MNPs to specific sites. The first study related to magnetic targeting was conducted by Meyers in 1963, who demonstrated that small iron particles injected intravenously into the leg veins of dogs could be accumulated using an external magnet Meyers et al. (1963). Later, in 1975, Turner and Rand built upon Gilchrist’s work and integrated this radiofrequency heating technique with embolization therapy Turner et al. (1975). These studies laid the foundation for research on the magnetic guidance of nanoparticles and magnetic hyperthermia, and they also inspired subsequent studies on drug targeting systems. The use of magnetic micro- and nanoparticles as therapeutic drug delivery carriers targeting specific areas of the body was first proposed in the late 1970s Senyei et al. (1978), Mosbach and Schröder (1979). Magnetic micro- and nanoparticles capable of binding cytotoxic drugs were developed by Widder and colleagues Widder et al. (1978). Lübbe et al. (1996) carried out the first clinical trials in humans on magnetic drug targeting, employing a ferrofluid (≈100 nm particle size) chemically attached to the anticancer drug epirubicin Lübbe et al. (1996). These studies are considered important milestones in the development of MDT applications.
Some of the anticancer drugs currently used in clinical practice have been successfully conjugated with magnetic components. Examples of studies in which magnetic nanoparticles (MNPs) are used in combination with anticancer drugs are presented in Table 2, along with their corresponding references. The table provides a non-exhaustive list of representative examples from the literature.
Table 2
|
Table 2 A Non-Exhaustive List of Studies İnvolving
the İntegrated Use of Magnetic Nanoparticles (MNPs and Anticancer Drugs Bilgili (2022) |
||
|
Antineoplastic
Drug Group |
MNP-Bound
Drug Name |
Reference |
|
Cytotoxic Antibiotics |
Doxorubicin |
Liang et al. (2016) , Munnier et al. (2008) |
|
(Anthracycline
Group Drugs) |
Mitoxantrone |
Heidari Majd et al. (2013), Krukemeyer et al. (2012) |
|
Epirubicin |
Xiong et al. (2017), Jalalian et al. (2013) |
|
|
Daunorubicin |
Wang et al. (2011), Lai et al. (2009) |
|
|
Idarubicin |
Gunduz et al. (2014) |
|
|
Monoclonal
Antibodies |
Trastuzumab |
Almaki
et al. (2017) |
|
Rituximab |
Song et al. (2020), Azadbakht et al. (2018) |
|
|
Cetuximab |
Kaluzova et al. (2015), Zang et al. (2019) |
|
|
Bevacizumab |
Lin et al. (2018), Savin et al. (2019) |
|
|
Taksan |
Paclitaxel |
Chorny et al. (2010), Tarantash et al. (2018) |
|
Docetaxel |
Ling et al. (2011), Panda et al. (2019) |
|
|
Pilatin |
Cisplatin |
Babincova et al. (2008), Toro-Cordova et al. (2018) |
|
Carboplatin |
Song et al. (2019), Davarpanah et al. (2018) |
|
|
Oxaliplatin |
Jabalera et al. (2019), Liu et al. (2018) |
|
|
Vinblastine |
Huang
et al. (2019), Albermani
et al. (2009) |
|
|
Vinca
Alkaloids and Analogues |
Vincristine |
Wu et al. (2016), Al-Musawi et al. (2021) |
|
Other Cytotoxic Antibiotics |
Bleomycin |
Kavaz et al. (2010), Xu et al. (2013) |
|
Mitomycin |
Ren et al. (2005), Yan et al. (2006) |
|
|
Pyrimidine Analogues |
Cytarabine |
Shabani et al. (2019) |
|
Fluorouracil |
Hashemi-Moghaddam et al. (2016), Ehi-Eromosele et al. (2017) |
|
|
Gemcitabine |
Parsian et al. (2016), Viota et al. (2013) |
|
|
Capecitabine |
Ghadiri et al. (2017), Afzali et al. (2020) |
|
|
Azacitidine |
Liu et al. (2010) |
|
|
Uracil |
Medine et al. (2011) |
|
|
Nitrogen Mustard Analogues |
Chlorambucil |
Yuan et al. (2010) |
|
Melphalan |
Shanmugavel and Karthikeyan
(2014) |
|
|
Ifosfamide |
Kong et al. (2015) |
|
|
Methotrexate |
Attari et al. (2019), Kohler et al. (2006) |
|
|
Folic Acid Analogues |
Pemetrexed Disodium |
Ak et al. (2020), Mohapatra et al. (2014) |
|
Imatinib |
Karimi Ghezeli et al. (2019), Dahiya and Dureja (2016) |
|
|
Protein Kinase Inhibitors |
Gefitinib |
Borg et al. (2020) |
In addition to the
antineoplastic drugs listed in Table 2, certain drugs such as tamoxifen, which is
widely used in hormone therapy for breast cancer as an adjuvant treatment to
prevent disease recurrence after breast surgery, have also been conjugated with
magnetic nanoparticles (MNPs) Majd et al. (2013).
Compared with conventional chemotherapy, targeted nanomedicines provide reduced non-specific cytotoxicity and can be engineered to exhibit multiple functionalities Such delivery systems are designed to transport adequate amounts of the relevant drug, often toxic, to specific regions of the body while shielding it from the surrounding biological environment . Drug targeting systems facilitate the use of more effective diagnostic and therapeutic strategies for improved disease management, while also contributing to the reduction of overall healthcare costs Rai and Morris (2019). A common problem encountered in cancer therapy is low specificity, meaning that the drugs used lack selectivity and destroy not only cancer cells but also all rapidly dividing cells in the body, including normal cells Pardee and Stein (2009). This situation leads to several challenges associated with chemotherapy treatment. From a clinical effectiveness perspective, the focus of anticancer drug development in recent years has shifted toward targeted drug delivery, due to limitations of cytotoxic therapies such as dose-limiting toxicity and drug resistance. To improve therapeutic outcomes without increasing toxicity, strategies aimed at overcoming drug resistance and inhibiting tumor growth should be relatively more specific to tumor cells than to normal tissues. Therefore, numerous strategies have been developed to achieve high therapeutic efficacy with reduced toxicity and/or overcome multidrug resistance, including the use of adjuvant or neoadjuvant therapies, combination therapies with other drugs and treatment modalities, and more recently, the use of specific and selective targeting approaches Pardee and Stein (2009), Baykara (2016). Cancer cells may fail to respond to one or more anticancer drugs, or they may initially respond to treatment but later develop resistance. Drug resistance in cells is primarily associated with mutations occurring in cancer cells. Reduced activity in membrane transport mechanisms or mutations in proteins responsible for mediating the intracellular action of drugs can lead to the development of drug resistance Ullah (2008). Such unfavorable outcomes that limit treatment efficacy have necessitated the development of new approaches for cancer therapy.
Since organs are located deep within the body cavity (around 8 ± 12 cm away from the body surface , concentrating the magnetic flux density on the tumor region region becomes significantly more complex. To overcome this challenge, two main approaches have been proposed: (a) the use of larger particles, or (b) the use of a stronger magnetic field Alexiou et al. (2000).
Magnetic Drug Targeting (MDT) studies generally involve two multidisciplinary components. The first focuses on the synthesis of magnetic nanoparticles (MNPs) and drug conjugation, while the second involves the transport and delivery of the drug-loaded nanoparticles. The synthesis of MNPs includes several stages such as selection of synthesis methods and their reproducibility, management of particle size and size distribution, stabilization of the synthesized particles, evaluation of toxicity, and surface coating with biocompatible and stealth materials to prolong systemic circulation in the body. The final step in this process involves drug conjugation to the nanoparticle surface. The drug transport component involves studies related to the generation of high-gradient magnetic fields, he interaction between the applied magnetic field and particle size , the transport profiles of MNPs within blood vessels, magnetohydrodynamic effects, blood rheology, and the physiology of cancer cells Bilgili (2022).
The difference between the success and failure of chemotherapy depends not only on the drug itself but also on how effectively it is delivered to its target. Due to the relatively non-specific effects of chemotherapeutic agents, even under optimal conditions there is almost always a certain degree of toxicity in normal tissues. Hence, the ability to accurately deliver the antineoplastic agent to the tumor site is critically important , as it can reduce systemic toxic side effects resulting from general systemic distribution and allow the use of much lower drug doses Alexiou et al. (2000). One of the major disadvantages of most chemotherapeutic agents is their lack of specificity. After intravenous administration, therapeutic drugs circulate systemically and are distributed across the body. As a result, the drugs affect not only the target tumor cells but also normal, healthy cells, causing harmful side effects. As an example, the toxic effects associated with anti-inflammatory drugs used in patients suffering from chronic arthritis may result in the discontinuation of therapy However, if these treatments could be directed to a specific site, such as an individual joint it would be possible to continue using these highly potent and effective agents without causing widespread systemic toxicity Pankhurst et al. (2003).
In magnetically guided therapy, a cytotoxic drug is linked
to a biocompatible magnetic nanoparticle carrier. The drug–carrier complexes
are administered intravenously and distributed through the circulatory system Upon
entering the bloodstream, the particles are guided by externally applied
high-gradient magnetic fields to accumulate at a specific target site in the
body Figure 2 Once localized at the
target region, the drug–carrier complex can release the drug through enzymatic
activity or changes in physiological factors such as osmolality, temperature or
pHor Alexiou et al. (2000), and can then be
internalized by tumor cells Pankhurst
et al. (2003).
Figure 2
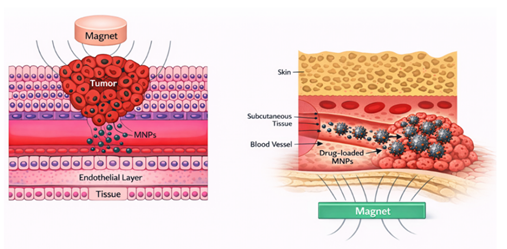
|
Figure 2 Cross-Sectional
Schematic Representation of a Hypothetical Magnetic Drug Delivery System Bilgili (2022) |
Magnetic drug delivery using particle carriers represents
a highly effective method for delivering therapeutic agents o a targeted
disease site. Very high levels of chemotherapeutic or radiological agents can
be concentrated near the target region, such as a tumor, without producing
toxic effects in nearby normal tissues or elsewhere in the body. Figure 3 demonstrates the
concept of magnetic targeting by contrasting systemic drug distribution with
magnetically guided delivery In magnetic drug targeting, a drug or therapeutic
radioisotope is conjugated to a magnetic compound, administered into the
patient’s bloodstream, and subsequently localized at the target site by a
strong magnetic field (see the arrow in Figure 3). Based on the type of
drug used, the therapeutic agent may either be gradually released from magnetic
carriers (e.g., chemotherapeutic drugs from MNPs) or induce a localized
therapeutic effect, such as irradiation from radioactive microspheres or hyperthermia
generated by magnetic nanoparticles. Consequently, large amounts of
systemically circulating drugs can be substituted with substantially smaller
doses specifically targeted to magnetically localized disease sites
makes it possible to achieve effective treatment with significantly
increased local drug concentrations, sometimes several times
higher than conventional systemic delivery Häfeli
and Pauer (1999).
Figure 3
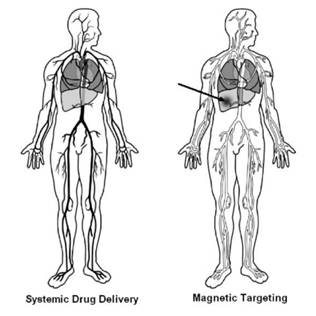
|
Figure 3 Concept of Magnetic Drug Targeting Häfeli (2004) |
The therapeutic effectiveness is influenced by several
physical parameters, such as magnetic field strength, magnetic field gradient,
and the volumetric and magnetic characteristics of the particles. Since the
carriers, namely ferrofluids, are generally administered through intravenous or
intra-arterial injection various flow-related
parameters also play an important role, such as blood flow velocity, circulation
time, the level of ferrofluid concentration, and route of
administration. In addition, physiological characteristics, including the depth
of the target tissue, defined as the distance from the magnetic field source , the
reversibility and binding strength between the drug and carrier, and tumor
volume also significantly influence the efficiency of the therapy Pankhurst et al. (2003), Lübbe et
al. (1996).
Barriers to Drug Targeting
Administered drugs
must first overcome the barriers encountered within the circulatory system .
The amount of drug that reaches the appropriate target region through
circulation subsequently encounters cellular barriers at the target site. As a
result, only a small fraction of the drug is internalized by the cells,
contributing to the therapeutic effect.
Following intravenous administration,
nanoparticles are quivkly surrounded by circulating molecules, including plasma
proteins once they enter the bloodstream. This process is termed opsonization
and significantly influences the fate of the injected particles Davis
(1997). Under normal
conditions, opsonization facilitates recognition of the particles by RES, which
constitutes the primary defense system of the body. The RES consists of
specialized phagocytic cells related to the connective tissue network
of organs such as the liver, spleen, and lymph nodes Kreuter
(1994). Kupffer cells in the liver, along with macrophages in the spleen
and circulating macrophages to a lesser extent, play an important role in the
removal of opsonized particles from the bloodstream. Physicochemical
characteristics that affect the opsonization process including particle size,
surface charge density, and the hydrophilic–hydrophobic balance have been
thoroughly examined in both liposomal and polymer-based nanoparticle systems Generally,
carrier surfaces that are smaller, more neutral, and more hydrophilic show
prolonged plasma half-lives. Considering the size effect, it has been suggested
that changes in surface curvature can affect the level and/or
type of opsonin adsorption Mornet
et al. (2004). In intravenous administration,
colloidal drug carriers are typically cleared from the bloodstream within a few
minutes, and their final biodistribution is generally 80–90% in the liver,
5–8% in the spleen, and 1–2% in the bone marrow Mornet
et al. (2004).
Consequently, the in vivo or ex vivo application of nanoparticles requires appropriate surface
functionalization to ensure that the particles remain non-toxic, biocompatible,
and stable against clearance by the reticuloendothelial system (RES) Berry
and Curtis (2003).
Figure 4
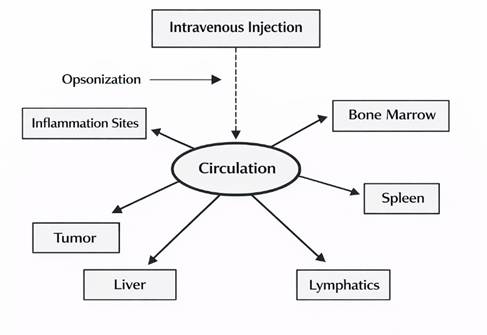
|
Figure 4 Following
Administration, the Particles are Quickly Coated by Plasma Proteins Through
Opsonization Berry et al. (2003) |
Nanoparticles with
hydrophobic surfaces are efficiently coated by plasma components and are
therefore quickly eliminated from circulation, whereas more hydrophilic
particles can resist this coating and are removed more slowly from the
bloodstream According to the literature, the most frequently used coatings
include dextran derivatives, polyethylene glycol, polyethylene oxide,
poloxamers, and polyoxamines Lacava et al. (2001). Dense polymer brushes serve to inhibit
opsonization, which allows nanoparticles to remain in the bloodstream for
longer periods Brigger
et al. (2012), Shen et al. (1996), Gref et al. (1994). Another strategy to
avoid uptake by the reticuloendothelial system is to minimize particle size Gref et al. (1994), Moghimi
et al. (2001). However, despite
these approaches, coated nanoparticles are still not able to completely avoid
uptake by the reticuloendothelial system Gaur et al. (2000) Berry
and Curtis (2003).
The major
challenges associated with systemic drug administration and the barriers that
anticancer drugs must overcome to reach cancer cells in vivo can be summarized
as follows Brigger
et al. (2012), Torchilin
(2000):
·
physiological
barriers (cell-independent mechanism),
·
cell-based
drug resistance mechanisms
·
resistance
to drug therapy resulting from the biodistribution, biotransformation, and
elimination of anticancer drugs
within the body,
·
lack of
drug-specific affinity toward pathological sites,
·
the
requirement for a large quantity of drug doses to accomplish the goal of sufficiently
elevated local concentrations, and
·
non-specific
toxic effects and other adverse reactions associated with dosages
The effectiveness of anticancer drugs can also be considerably influenced depending on the route of administration. For instance, orally administered therapeutics may lose activity in acidic environments or undergo absorption and metabolism in the liver and gastrointestinal tract, which can significantly reduce systemic bioavailability. When nanoparticles larger than 10 nm are administered intravenously (i.v.), they may lead to non-specific deposition of therapeutic agents in the reticuloendothelial system of the liver and spleen , which reduces the quantity of circulating therapeutic agents in the body. In contrast, therapeutic agents with sizes smaller than 5 nm, similar to small-molecule drugs, can be rapidly cleared from the bloodstream through renal and hepatic elimination. Interactions between therapeutic agents and serum proteins can influence their biodistribution, pharmacokinetic behavior, bioactivity, and targeting efficiency of therapeutic agents and nanoparticle drug. Large macromolecules such as proteins and antibodies, as well as viral vectors and nanoparticles are not readily cleared by the kidneys and thus display longer circulation times in the bloodstream, allowing greater delivery of the drug to tumor sites. The bioavailability of nanoparticle-based therapeutics is also affected by macrophages. However, when the size of the therapeutic agent exceeds approximately 200–400 nm, the particles can be rapidly cleared from circulation due to enhanced macrophage uptake. One of the most important challenges in chemotherapy often causes severe systemic toxicity, which restricts the maximum dose that can be given to patients and often leads to harmful side effects that affect patient health and well-being. In addition, small-molecule chemotherapeutic drugs are rapidly cleared from the bloodstream, typically exhibiting short circulation half-lives of approximately 15–30 minutes. Drug specificity can be improved by delivering therapeutics to tumors using antibody-based targeting strategies. Due to their relatively large size, antibody-based therapeutics generally have longer half-lives and higher delivery efficiency to tumors compared with conventional chemotherapeutic drugs. Therapeutic agents may also be administered via nanoparticle-based delivery systems. Nanoparticle drug carriers, typically smaller than 100–200 nm, can accumulate in tumor tissues via the enhanced permeability and retention EPR effect, which allows nanoparticles to pass through leaky tumor vasculature and accumulate in the perivascular region (the area surrounding blood vessels) Rai and Morris (2019).
Major Challenges Associated with Magnetic Drug Targeting
The major problems related to systemic drug administration include the biological distribution the systemic distribution of pharmaceuticals, the lack of specificity toward pathological sites, the need for high drug doses to obtain adequate local concentrations, and non-specific toxicity with additional adverse effects. Targeted drug delivery strategies seek to overcome many of these limitations Torchilin (2000). One of the fundamental approaches to drug targeting is magnetic targeting, which involves guiding a drug immobilized on magnetic materials using an external magnetic field Berry and Curtis (2003).
For magnetic drug targeting (MDT) to be successful, it must be both safe and effective, meaning that the maximum therapeutic effect should be achieved with a minimal amount of magnetic particles, enabling the drug to be efficiently delivered and transported to the target site.
From a biophysiological perspective, several parameters
related to the ferrofluid-bound drug complex and the applied
magnetic field plays a critical role including:
1)
size of
the particles
2)
surface properties of the nanoparticles,
3)
ferrofluid particle concentration,
4)
properties of the carrier fluid,
5)
the reversibility and binding strength between
the drug and ferrofluid (desorption properties)
6)
administration route to the organism
7)
injection/infusion dutation time and rate,
8)
the magnetic field configuration and strength
9)
He time period of magnetic field application
The patient’s
physiological parameters also play an important role and include:
1)
body size, body weight, and body surface area
2)
the volume of circulating blood
3)
cardiac output and overall vascular resistance,
4)
blood circulation time, (etumor dimensions and anatomical location
5)
tumor
volume and location,
6)
vascularization of the tumor, and
7)
blood flow within the tumor.
Since
Physiological parameters are influenced by the size of the organism, the
type
of the characteristics of the ferrofluid–drug complex, the type of the
characteristics of the ferrofluid–drug complex parameters for the effectiveness
of MDT are the ferrofluid bioavailability
and the in the in vivo drug release time Lübbe et al. (1996).
To understand this
new pharmacological approach and its mechanism of action, several factors must
be considered, which can be grouped into different categories. First, there are
ferrofluid-related parameters, including the particle dimensions, surface
characteristics, the concentration and volume of the carrier, together
with magnetic field strength, which influence the characteristics of the
drug–particle interaction. Second, there are factors related to delivery into
the organism, including the pathway of administration of administration,
as well as the injection rate and duration or infusion.
Finally, the physiological characteristics of the organism must be considered,
encompassing factors such as body weight, blood volume, cardiac output,
circulation time, tumor volume, tumor location, and tumor blood flowLübbe et al. (1996), Lübbe et al. (1996). Therefore, moving from
animal experiments to human clinical trials is a complex process and presents
significant challenges Berry
and Curtis (2003).
Several challenges
are related to magnetically targeted drug delivery Pankhurst
et al. (2003), Tartaj
et al. (2003), Lübbe et
al. (1996), Häfeli
(2004). These challenges include:
1)
The risk
of blood vessel embolization at the target site caused by the accumulation of
magnetic carrier,
2)
Challenges
associated with scaling up from animal models, resulting from the larger
distance between the target site and the external magnet,
3)
following
release, the drug is no longer guided by the magnetic field,
4)
potential
adverse toxic effects of magnetic carriers,
5)
forces
acting on the particles within the bloodstream,
6)
magnetic
forces created by the externally applied magnet,
7)
Concentration
of magnetic nanoparticles (MNPs),
8)
Tumor
volume,
9)
Reversibility
of the drug–carrier binding,
10) Tissue depth,
11) Circulation time, and
12) Route of administration (e.g., intravenous
(i.v.) or intra-arterial (i.a.)).
Nevertheless, recent
experimental and preclinical research suggests that these limitations may still
be addressed, improve drug retention through magnetic targeting, and
simultaneously address potential safety concerns Pardee
and Stein (2009), Lübbe e
t al. (1996).
Conclusion
The treatment of cancer, primarily with chemotherapy, has proven to be difficult and often not only has poor selectivity but also has serious side effects. Chemotherapeutic drugs are administered in a systemic manner, through the circulatory system, damaging healthy cells in the body and is a key contributor to the limited efficacy of therapy. As the field of cancer research has emphasized controlled drug delivery systems in recent years, the design of such systems has increasingly been important. Due to their small size, high surface-to-volume ratio and controllability using external magnetic fields, magnetic nanoparticles have gained increasing importance in biomedical applications. Notably, nanoparticles based on iron oxides, such as magnetite (Fe₃O₄) and maghemite (γ-Fe₂O₃), are frequently employedin magnetic drug targeting applications due to their biocompatibility and superparamagnetic character. The functionalization of such nanoparticles with biocompatible polymers or inorganic coatings yields stability in biological environments, thereby making their surface friendly to the application of anticancer drugs. MDT (Magnetic Drug Targeting) technique enables the targeted application of drugs loaded with to the targeted sites of tumor with assistance of an external magnetic field. By this approach, high concentrations of drug can reach the target site and systemic toxicity and side effects are diminished to an extremely small extent. Magnetic NPs not only represent drug delivery devices but also find applications in other several biomedical applications, including magnetic resonance imaging (MRI), magnetic hyperthermia, biosensors, gene therapy, and tissue engineering But numerous technical and biological obstacles need to be surpassed to ensure the translation of magnetic drug-targeting technology into clinical application. Specifically, the feasibility of nanoparticle biocompatibility, toxicity, rapid clearance by the reticuloendothelial system, penetration of magnetic fields into deep tissues and amount of nanoparticle accumulation at the specific target are issues that need ongoing research. These findings demonstrate the remarkable potential of magnetic nanoparticle-based drug delivery systems that could lead to the development of targeted and more effective therapeutic strategies to address the issues of cancer. Thanks to the latest trends in nanotechnology, materials science, and biomedical engineering, it is anticipated that using magnetic drug targeting systems can play an increasingly important role in producing safer, more effective, personalized treatments for cancer.
ACKNOWLEDGMENTS
None.
REFERENCES
Afzali, M., Mostafavi, A., and Shamspur, T. (2020). A Novel Electrochemical Sensor Based on Magnetic Core@Shell Molecularly Imprinted Nanocomposite for Sensitive and Selective Determination of Anticancer Drug Capecitabine. Arabian Journal of Chemistry, 13(8), 6626-6638. https://doi.org/10.1016/j.arabjc.2020.06.018
Ak, G., Aksu, D., Çapkın, E., Sarı, Ö., Kımız Geboloğlu, I., and Şanlıer, Ş. H. (2020). Delivery of Pemetrexed by Magnetic Nanoparticles: Design, Characterization, in Vitro and in Vivo Assessment. Preparative Biochemistry & Biotechnology, 50(3), 215-225. https://doi.org/10.1080/10826068.2019.1692220
Al-Musawi, S., et al. (2021). Smart Nanoformulation Based on Polymeric Magnetic Nanoparticles and Vincristine Drug: A Novel Therapy for Apoptotic Gene Expression in Tumors. Life, 11(1), 71. https://doi.org/10.3390/life11010071
Albermani, M. S., Al-Alwani, A. J., and Al, Z. A. (2009). Vinblastine Based Iron Oxide Nano
Drug Delivery System.
Alexiou,
C., Jurgons, R., Schmid, R. J., Hilpert, A., Bergemann, C., Parak, F. G., and
Iro, H. (2000).
Locoregional Cancer Treatment with Magnetic Drug Targeting. Cancer Research,
60(23), 6641-6648.
Alexiou, C., Jurgons, R., Schmid, R. J., Hilpert, A., Bergemann, C., Parak, F. G., and Iro, H. (2000). Locoregional cancer treatment with magnetic drug targeting. Cancer Research, 60(23), 6641-6648.
Alexiou, C., Schmid, R. J., Jurgons, R., Kremer, M., Wanner, G., Bergemann, C., Huenges, E., Nawroth, T., Arnold, W., and Parak, F. G. (2011). Cancer Therapy with Drug Loaded Magnetic Nanoparticles-Magnetic Drug Targeting. Journal of Magnetism and Magnetic Materials, 323(10), 1404-1407. https://doi.org/10.1016/j.jmmm.2010.11.059
Alexiou, C.,
Schmidt, A., Klein, R., Hulin, P., Bergemann, C., and Arnold, W. (2002). Magnetic Drug Targeting:
Biodistribution and Dependency on Magnetic Field Strength. Journal of Magnetism
and Magnetic Materials, 252, 363-366. https://doi.org/10.1016/S0304-8853(02)00605-4
Almaki, J. H., Nasiri, R., Idris, A., Nasiri, M., Majid, F. A. A., and Losic, D. (2017). Trastuzumab-Decorated Nanoparticles for in Vitro and in vivo Tumor-Targeting Hyperthermia of HER2+ Breast Cancer. Journal of Materials Chemistry B, 5(35), 7369-7383. https://doi.org/10.1039/C7TB01305A
Angelakeris, M. (2017). Magnetic Nanoparticles: A Multifunctional Vehicle for Modern Theranostics. Biochimica et Biophysica Acta (BBA) - General Subjects, 1861(6), 1642-1651. https://doi.org/10.1016/j.bbagen.2017.02.022
Antfolk, M., and Laurell, T. (2017). Continuous Flow Microfluidic Separation and Processing of Rare Cells and Bioparticles Found in Blood-A Review. Analytica Chimica Acta, 965, 9-35. https://doi.org/10.1016/j.aca.2017.02.017
Attari, E., Nosrati, H., Danafar, H., and Kheiri Manjili, H. (2019). Methotrexate Anticancer Drug Delivery to Breast Cancer Cell Lines by Iron Oxide Magnetic Based Nanocarrier. Journal of Biomedical Materials Research Part A, 107(11), 2492-2500. https://doi.org/10.1002/jbm.a.36755
Azadbakht, B., Afarideh, H., Ghannadi-Maragheh, M., Bahrami-Samani, A., and Yousefnia, H. (2018). Absorbed Doses in Humans from 188Re-Rituximab in the free form and Bound to Superparamagnetic Iron Oxide Nanoparticles: Biodistribution Study in mice. Applied Radiation and Isotopes, 131, 96-102. https://doi.org/10.1016/j.apradiso.2017.10.041
Babincova, M., Altanerova, V., Bergemann, C., and Babinec, P. (2008). In Vitro Analysis of Cisplatin Functionalized Magnetic Nanoparticles in Combined Cancer Chemotherapy and Electromagnetic Hyperthermia. IEEE Transactions on Nanobioscience, 7(1), 15-19. https://doi.org/10.1109/TNB.2008.2000145
Babincova, M., Sourivong, P., Leszczynska, D., and Babinec, P. (2000). Blood-Specific Whole-Body Electromagnetic Hyperthermia. Medical Hypotheses, 55(6), 459-460. https://doi.org/10.1054/mehy.2000.1089
Baykara, O. (2016). Kanser Tedavisinde Güncel Yaklaşımlar. Balıkesir Sağlık Bilimleri Dergisi, 5(3), 154-165.
Berry, C. C., and Curtis, A. S. G. (2003). Functionalisation of Magnetic Nanoparticles for Applications In Biomedicine. Journal Of Physics D: Applied Physics, 36(13), R198. https://doi.org/10.1088/0022-3727/36/13/203
Bilgili, H. (2022). Manyetik Nanoparçacıklar Içeren Kan Imitasyon Malzemelerinde Manyetik Ilaç Hedefleme Işlemlerinin Incelenmesi [Doctoral Dissertation, Inonu University].
Bohara, R. A., Thorat, N. D., and Pawar, S. H. (2016). Role of functionalization: Strategies to Explore Potential Nano-Bio Applications of Magnetic Nanoparticles. RSC Advances, 6(50), 43989-44012. https://doi.org/10.1039/C6RA02129H
Borg, H., et al. (2020). Preconcentration and Detection of Gefitinib Anti-Cancer Drug Traces from Water and Human Plasma Samples by Means of Magnetic Nanoparticles. Nanomaterials, 10(6), 1196. https://doi.org/10.3390/nano10061196
Boutry, S., Laurent, S., Vander Elst, L., and Muller, R. N. (2006). Specific E-Selectin Targeting with a Superparamagnetic MRI Contrast Agent. Contrast Media & Molecular Imaging, 1(1), 15-22. https://doi.org/10.1002/cmmi.87
Brigger, I., Dubernet, C., and Couvreur, P. (2012). Nanoparticles in Cancer Therapy and Diagnosis. Advanced Drug Delivery Reviews, 64, 24-36. https://doi.org/10.1016/j.addr.2012.09.006
Canefe,
K., and Duman, G. (1994). Selective Drug Delivery and Targeting. Ankara Üniversitesi
Eczacılık Fakültesi Dergisi, 23(1), 53-63.
Cartmell, S., Dobson, J., Verschueren, S., Hughes, S., and El Haj, A. (2002). Mechanical Conditioning of Bone Cells in Vitro Using Magnetic Microparticle Technology. European Cells and Materials, 4, 42-44.
Carvalho, C., Santos, R. X., Cardoso, S., Correia, S., Oliveira, P. J., Santos, M. S., and Moreira, P. I. (2009). Doxorubicin: The Good, the Bad and the Ugly Effect. Current Medicinal Chemistry, 16(25), 3267-3285. https://doi.org/10.2174/092986709788803312
Chan, D. C., Kirpotin, D. B., and Bunn, P. A., Jr. (1993). Synthesis and Evaluation of Colloidal Magnetic Iron Oxides for the Site-Specific Radiofrequency-Induced Hyperthermia of Cancer. Journal of Magnetism and Magnetic Materials, 122(1-3), 374-378. https://doi.org/10.1016/0304-8853(93)91113-L
Chorny, M., Fishbein, I., Yellen, B. B., Alferiev, I., Bakay, M., Ganta, S., Adamo, R., De Rosa, G., and Levy, R. J. (2010). Targeting Stents with Local Delivery of Paclitaxel-Loaded Magnetic Nanoparticles Using Uniform Fields. Proceedings of the National Academy of Sciences, 107(18), 8346-8351. https://doi.org/10.1073/pnas.0909506107
Chouly, C., Pouliquen, D., Lucet, I., Jeune, J., adn Jallet, P. (1996). Development of Superparamagnetic Nanoparticles for MRI: Effect of Particle Size, Charge and Surface Nature on Biodistribution. Journal of Microencapsulation, 13(3), 245-255. https://doi.org/10.3109/02652049609026013
Chourpa, I., et al. (2005). Molecular Composition of Iron Oxide Nanoparticles, Precursors for Magnetic Drug Targeting, as Characterized by Confocal Raman Microspectroscopy. Analyst, 130(10), 1395-1403. https://doi.org/10.1039/b419004a
Corot, C., Robert, P., Idée, J.-M., and Port, M. (2006). Recent Advances in Iron Oxide Nanocrystal Technology for Medical Imaging. Advanced Drug Delivery Reviews, 58(14), 1471-1504. https://doi.org/10.1016/j.addr.2006.09.013
Dahiya, M., and Dureja, H. (2016). Central Composite Designed Imatinib-Loaded Magnetic Nanoparticles. Current Nanomedicine, 6(2), 146-155. https://doi.org/10.2174/2468187306666160802125718
Dailey, J., Phillips, J., Li, C., and Riffle, J. (1999). Synthesis of Silicone Magnetic Fluid for use in Eye Surgery. Journal of Magnetism and Magnetic Materials, 194(1-3), 140-148. https://doi.org/10.1016/S0304-8853(98)00562-9
Davarpanah, F., Yazdi, A. K., Barani, M., Mirzaei, M., and Torkzadeh-Mahani, M. (2018). Magnetic Delivery of Antitumor Carboplatin by Using Pegylated-Niosomes. DARU Journal of Pharmaceutical Sciences, 26(1), 57-64. https://doi.org/10.1007/s40199-018-0215-3
Dave, S. R., and Gao, X. (2009). Monodisperse Magnetic Nanoparticles for Biodetection, Imaging, and Drug Delivery: A Versatile and Evolving Technology. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology, 1(6), 583-609. https://doi.org/10.1002/wnan.51
Davis, S. (1997). Biomedical Applications of Nanotechnology-Implications for Drug Targeting and Gene Therapy. Trends in Biotechnology, 15(6), 217-224. https://doi.org/10.1016/S0167-7799(97)01036-6
Deng, Y., Wang, L., Yang, W., Fu, S., and Elaïssari, A. (2003). Preparation of Magnetic Polymeric Particles Via Inverse Microemulsion Polymerization Process. Journal of Magnetism and Magnetic Materials, 257(1), 69-78. https://doi.org/10.1016/S0304-8853(02)00987-3
Dobson, J., Keramane, A., and El Haj, A. (2002). Theory and Applications of a Magnetic Force Bioreactor. European Cells and Materials, 4(Suppl. 2), 42-44.
Durán, J. D., Arias, J. L., Gallardo, V., and Delgado, A. V. (2008). Magnetic Colloids as Drug Vehicles. Journal of Pharmaceutical Sciences, 97(8), 2948-2983. https://doi.org/10.1002/jps.21249
Ehi-Eromosele, C., Ita, B., and Iweala, E. (2017). Silica Coated LSMO MagnEtic Nanoparticles for the pH-Responsive Delivery of 5-Fluorouracil Anticancer Drug. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 530, 164-171. https://doi.org/10.1016/j.colsurfa.2017.07.059
Gaur, U., Sahoo, S. K., De, T., Ghosh, P. C., Maitra, A., and Ghosh, P. (2000). Biodistribution of Fluoresceinated Dextran Using Novel Nanoparticles Evading Reticuloendothelial System. International Journal of Pharmaceutics, 202(1-2), 1-10. https://doi.org/10.1016/S0378-5173(99)00447-0
Ghadiri, M., Vasheghani-Farahani, E., Atyabi, F., Kobarfard, F., and Hosseinkhani, H. (2017). In-Vitro Assessment of Magnetic Dextran-Spermine Nanoparticles for Capecitabine Delivery to Cancerous Cells. Iranian Journal of Pharmaceutical Research, 16(4), 1320.
Gilchrist, R. K., Medal, R., Shorey, W. D., Hanselman, R. C., Parrott, J. C., and Taylor, C. B. (1957). Selective inductive heating of Lymph Nodes. Annals of Surgery, 146(4), 596. https://doi.org/10.1097/00000658-195710000-00007
Grasset, F., Labhsetwar, N., Li, D., Park, D. C., Saito, N., Haneda, H., Roisnel, T., Mornet, S., Duguet, E., Portier, J., and Etourneau, J. (2001). Synthesis, Magnetic Properties, Surface Modification and Cytotoxicity Evaluation of Y3Fe5−xAlxO12 (0 ≤ x ≤ 2) Garnet Submicron Particles for Biomedical applications. Journal of Magnetism and Magnetic Materials, 234(3), 409-418. https://doi.org/10.1016/S0304-8853(01)00386-9
Gref, R., Minamitake, Y., Peracchia, M. T., Trubetskoy, V., Torchilin, V., and Langer, R. (1994). Biodegradable long-circulating polymeric nanospheres. Science, 263(5153), 1600-1603. https://doi.org/10.1126/science.8128245
Gunduz, U., Yalcin, S., Kayabasi, C., Okutan, H., Gacar, G., Sayitoglu, M., Piskin, E., and Aydin, R. S. T. (2014). Idarubicin-Loaded Folic Acid Conjugated Magnetic Nanoparticles as a Targetable Drug Delivery System for Breast Cancer. Biomedicine & Pharmacotherapy, 68(6), 729-736. https://doi.org/10.1016/j.biopha.2014.08.013
Gupta, A. K., and Gupta, M. (2005). Synthesis and Surface Engineering of Iron Oxide Nanoparticles for Biomedical Applications. Biomaterials, 26(18), 3995-4021. https://doi.org/10.1016/j.biomaterials.2004.10.012
Gupta, S., Alargova, R. G., Kilpatrick, P. K., and Velev, O. D. (2008). On-Chip Electric Field Driven Assembly of Biocomposites from Live Cells and Functionalized Particles. Soft Matter, 4(4), 726-730. https://doi.org/10.1039/b717850f
Hashemi-Moghaddam, H., Kazemi-Bagsangani, S., Jamili, M., and Zavareh, S. (2016). Evaluation of Magnetic Nanoparticles Coated by 5-Fluorouracil Imprinted Polymer for Controlled Drug Delivery in Mouse Breast Cancer Model. International Journal of Pharmaceutics, 497(1-2), 228-238. https://doi.org/10.1016/j.ijpharm.2015.11.040
Heidari Majd, M., Asgari, D., Barar, J., Valizadeh, H., Kafil, V., Abadpour, A., Mouhammadi, M., Rafi, M. A., and Omidi, Y. (2013). Specific Targeting of Cancer Cells by Multifunctional Mitoxantrone-Conjugated Magnetic Nanoparticles. Journal of Drug Targeting, 21(4), 328-340. https://doi.org/10.3109/1061186X.2012.750325
Huang, K.S., Yang, C.H., Wang, Y.C., Wang, W.T., and Lu, Y.-Y. (2019). Microfluidic Synthesis of Vinblastine-Loaded Multifunctional Particles for Magnetically Responsive Controlled Drug Release. Pharmaceutics, 11(5), 212. https://doi.org/10.3390/pharmaceutics11050212
Häfeli, U. (2004). Magnetically Modulated Therapeutic Systems. International Journal of Pharmaceutics, 277(1-2), 19-24. https://doi.org/10.1016/j.ijpharm.2003.03.002
Häfeli, U. O., and Pauer, G. J. (1999). In Vitro and in Vivo Toxicity of Magnetic Microspheres. Journal of Magnetism and Magnetic Materials, 194(1-3), 76-82. https://doi.org/10.1016/S0304-8853(98)00560-5
Jabalera, Y., Sola-Leyva, A., Carrasco-Jiménez, M. P., Iglesias, G. R., Jimenez-Lopez, C., and Marchal, J. A. (2019). Oxaliplatin-Biomimetic Magnetic Nanoparticle Assemblies for Colon Cancer-Targeted Chemotherapy: An in vitro study. Pharmaceutics, 11(8), 395. https://doi.org/10.3390/pharmaceutics11080395
Jain, T. K., Morales, M. A., Sahoo, S. K., Leslie-Pelecky, D. L., and Labhasetwar, V. (2005). Iron Oxide Nanoparticles For Sustained Delivery of Anticancer Agents. Molecular Pharmaceutics, 2(3), 194-205. https://doi.org/10.1021/mp0500014
Jalalian, S. H., Taghdisi, S. M., Shahidi Hamedani, N., Kalat, S. A., Lavaee, P., Zandkarimi, M., Ghows, N., and Jaafari, M. R. (2013). Epirubicin Loaded Super Paramagnetic Iron Oxide Nanoparticle-Aptamer Bioconjugate for Combined Colon Cancer Therapy and Imaging in Vivo. European Journal of Pharmaceutical Sciences, 50(2), 191-197. https://doi.org/10.1016/j.ejps.2013.06.015
Jordan, A., et al. (1996). Cellular Uptake of Magnetic Fluid Particles and their Effects on Human Adenocarcinoma Cells Exposed to AC Magnetic Fields in Vitro. International Journal of Hyperthermia, 12(6), 705-722. https://doi.org/10.3109/02656739609027678
Jung, C. W., and Jacobs, P. (1995). Physical and Chemical Properties of Superparamagnetic Iron Oxide MR Contrast Agents: Ferumoxides, Ferumoxtran, Ferumoxsil. Magnetic Resonance Imaging, 13(5), 661-674. https://doi.org/10.1016/0730-725X(95)00024-B
Kaluzova, M., Bouras, A., Machaidze, R., and Hadjipanayis, C. G. (2015). Targeted Therapy of Glioblastoma Stem-like cells and Tumor Non-Stem Cells using Cetuximab-Conjugated Iron-Oxide Nanoparticles. Oncotarget, 6(11), 8788. https://doi.org/10.18632/oncotarget.3554
Karimi Ghezeli, Z., Hekmati, M., and Veisi, H. (2019). Synthesis of Imatinib-Loaded Chitosan-Modified Magnetic Nanoparticles as an Anti-Cancer Agent for pH Responsive Targeted Drug Delivery. Applied Organometallic Chemistry, 33(4), e4833. https://doi.org/10.1002/aoc.4833
Kavaz, D., Odabaş, S., Güven, E., Demirbilek, M., and Denkbaş, E. B. (2010). Bleomycin Loaded Magnetic Chitosan Nanoparticles as Multifunctional Nanocarriers. Journal of Bioactive and Compatible Polymers, 25(3), 305-318. https://doi.org/10.1177/0883911509360735
Kohler, N., Sun, C., Fichtenholtz, A., Gunn, J., Fang, C., and Zhang, M. (2006). Methotrexate-Immobilized Poly(Ethylene Glycol) Magnetic Nanoparticles for MR Imaging and Drug Delivery. Small, 2(6), 785-792. https://doi.org/10.1002/smll.200600009
Kong, L., Hu, J., Qin, D., and Yan, P. (2015). Interaction of Ifosfamide-Loaded Superparamagnetic Iron Oxide Nanoparticles with Human Serum Albumin-A Biophysical Study. Journal of Pharmaceutical Innovation, 10(1), 13-20. https://doi.org/10.1007/s12247-014-9199-9
Kreuter, J. (1994). Drug Targeting with Nanoparticles. European Journal of Drug Metabolism and Pharmacokinetics, 19(3), 253-256. https://doi.org/10.1007/BF03188928
Krukemeyer, M. G., Krenn, V., Jakobs, M., and Wagner, W. (2012). Mitoxantrone-Iron Oxide Biodistribution in Blood, Tumor, Spleen, and Liver-Magnetic Nanoparticles in Cancer Treatment. Journal of Surgical Research, 175(1), 35-43. https://doi.org/10.1016/j.jss.2011.01.060
Kuznetsov, A. A., Yunin, A. M., Savichev, A. A., Kuznetsov, O. A., Dmitriev, N. S., and Palchun, V. T. (2001). Application of Magnetic Devices in Otiatria. Journal of Magnetism and Magnetic Materials, 225(1-2), 202-208. https://doi.org/10.1016/S0304-8853(00)01259-2
Lacava, L. M., et al. (2001). Magnetic Resonance of a Dextran-Coated Magnetic Fluid Intravenously Administered in mice. Biophysical Journal, 80(5), 2483-2486. https://doi.org/10.1016/S0006-3495(01)76217-0
Lai, B.-B., Chen, B.-A., Wu, Z.-X., Wang, J., Sheng, S.-L., Wang, X.-M., Liu, J., and Zhu, X.-Y. (2009). Daunorubicin-loaded Magnetic Nanoparticles of Fe(3)O(4) greatly enhance the Responses of Multidrug-Resistant K562 leukemic cells in a Nude Mouse Xenograft Model to Chemotherapy. Zhongguo Shi Yan Xue Ye Xue Za Zhi, 17(2), 345-351.
Laurent, S., Forge, D., Port, M., Roch, A., Robic, C., Vander Elst, L., and Muller, R. N. (2008). Magnetic Iron Oxide Nanoparticles: Synthesis, Stabilization, Vectorization, Physicochemical Characterizations, and Biological Applications. Chemical Reviews, 108(6), 2064-2110. https://doi.org/10.1021/cr068445e
Liang, P.C., Chen, Y.C., Chiang, C.F., Mo, L.R., Wei, S.Y., Hsieh, W.-Y., Lin, W.-L., and Doxorubicin-Modified Magnetic Nanoparticle Study Group. (2016). Doxorubicin-Modified Magnetic Nanoparticles as a Drug Delivery System for Magnetic Resonance Imaging-Monitoring Magnet-Enhancing Tumor Chemotherapy. International Journal of Nanomedicine, 11, 2021. https://doi.org/10.2147/IJN.S94139
Lin, R., Huang, J., Wang, L., Li, Y., Chang, Y., Li, W., and Shen, B. (2018). Bevacizumab and Near Infrared Probe Conjugated Iron Oxide Nanoparticles for Vascular Endothelial Growth Factor Targeted MR and Optical Imaging. Biomaterials Science, 6(6), 1517-1525. https://doi.org/10.1039/C8BM00225H
Ling, Y., Wei, K., Luo, Y., Gao, X., and Zhong, S. (2011). Dual Docetaxel/Superparamagnetic Iron Oxide Loaded Nanoparticles for Both Targeting Magnetic Resonance Imaging and Cancer Therapy. Biomaterials, 32(29), 7139-7150. https://doi.org/10.1016/j.biomaterials.2011.05.089
Liu, D., Wu, W., Ling, J., Wen, S., Gu, N., and Zhang, X. (2018). Target-Specific Delivery of Oxaliplatin to HER2-Positive Gastric Cancer Cells in Vivo Using Oxaliplatin-Au-Fe3o4-Herceptin Nanoparticles. Oncology Letters, 15(5), 8079-8087. https://doi.org/10.3892/ol.2018.8323
Liu, D., Wu, W., Ling, J., Wen, S., Gu, N., and Zhang, X. (2018). Target-Specific Delivery of Oxaliplatin to Her2-Positive Gastric Cancer Cells in Vivo Using Oxaliplatin-Au-Fe3o4-Herceptin Nanoparticles. Oncology Letters, 15(5), 8079-8087. https://doi.org/10.3892/ol.2018.8323
Liu, Y.-H., Guo, L., He, M.-C., and Shen, Z.-Y. (2010). Differentiation of Superparamagnetic Iron Oxide-Labeled Bone Marrow Mesenchymal Stem Cells into Cardiomyocytes-Like Cells in Vitro Induced By 5-azacitidine. Chinese Journal of Tissue Engineering Research, 14(40), 7415.
Lübbe, A. S., Alexiou, C., and Bergemann, C. (2001). Clinical Applications of Magnetic Drug Targeting. Journal of Surgical Research, 95(2), 200-206. https://doi.org/10.1006/jsre.2000.6030
Lübbe, A. S., Bergemann, C., Brock, J., and McClure, D. G. (1999). Physiological Aspects in Magnetic Drug-Targeting. Journal of Magnetism and Magnetic Materials, 194(1-3), 149-155. https://doi.org/10.1016/S0304-8853(98)00574-5
Lübbe, A. S., Bergemann, C., Huhnt, W., Fricke, T., Riess, H., Brock, J. W., and Huhn, D. (1996). Preclinical Experiences with Magnetic Drug Targeting: Tolerance and Efficacy. Cancer Research, 56(20), 4694-4701.
Lübbe, A. S., and Bergemann, C. (1997). Selected Preclinical and First Clinical Experiences with Magnetically Targeted 4′-Epidoxorubicin in Patients with Advanced Solid Tumors. In Scientific and Clinical Applications of Magnetic Carriers (457-480). Springer. https://doi.org/10.1007/978-1-4757-6482-6_35
Majd, M. H., et al. (2013). Tamoxifen Loaded Folic Acid Armed PEGylated Magnetic Nanoparticles for Targeted Imaging and Therapy of Cancer. Colloids and Surfaces B: Biointerfaces, 106, 117-125. https://doi.org/10.1016/j.colsurfb.2013.01.051
Medine, E. I., Ünak, P., Sakarya, S., and Özkaya, F. (2011). Investigation of in Vitro Efficiency of Magnetic Nanoparticle-Conjugated 125I-uracil Glucuronides in Adenocarcinoma Cells. Journal of Nanoparticle Research, 13(10), 4703-4715. https://doi.org/10.1007/s11051-011-0436-6
Meyers, P. H., Cronic, F., and Nice, C., Jr. (1963). Experimental Approach in the use and Magnetic Control of Metallic Iron Particles in the Lymphatic and Vascular System of Dogs as a Contrast and Isotropic Agent. American Journal of Roentgenology, Radium Therapy, and Nuclear Medicine, 90.
Mijailovich, S. M., Kojic, M., Zivkovic, M., Fabry, B., and Fredberg, J. J. (2002). A Finite Element Model of Cell Deformation During Magnetic Bead twisting. Journal of Applied Physiology, 93(4), 1429-1436. https://doi.org/10.1152/japplphysiol.00255.2002
Miller, M., et al. (2001). A DNA Array Sensor Utilizing Magnetic Microbeads and Magnetoelectronic Detection. Journal of Magnetism and Magnetic Materials, 225(1-2), 138-144. https://doi.org/10.1016/S0304-8853(00)01242-7
Moghimi, S. M., Hunter, A. C., and Murray, J. C. (2001). Long-Circulating and Target-Specific Nanoparticles: Theory to Practice. Pharmacological Reviews, 53(2), 283-318. https://doi.org/10.1016/S0031-6997(24)01494-7
Mohapatra, S., Rout, S. R., Narayan, R., and Maiti, T. K. (2014). Multifunctional Mesoporous Hollow Silica Nanocapsules for Targeted Co-Delivery of Cisplatin-Pemetrexed and MR Imaging. Dalton Transactions, 43(42), 15841-15850. https://doi.org/10.1039/C4DT02144D
Mornet, S., Grasset, F., Portier, J., and Duguet, E. (2002). Maghemite@ Silica Nanoparticles for Biological Applications. eCells and Materials Journal, 3(Suppl. 2), 110-113.
Mornet, S., Vasseur, S., Grasset, F., and Duguet, E. (2004). Magnetic Nanoparticle Design for Medical Diagnosis and Therapy. Journal of Materials Chemistry, 14(14), 2161-2175. https://doi.org/10.1039/b402025a
Mosbach, K., and Schröder, U. (1979). Preparation and Application of Magnetic Polymers for Targeting of Drugs. FEBS Letters, 102(1), 112-116. https://doi.org/10.1016/0014-5793(79)80940-0
Munnier, E., Cohen-Jonathan, S., Linassier, C., Douziech-Eyrolles, L., Marchais, H., Soucé, M., Hervé, K., Dubois, P., Chourpa, I., and Lucet, I. (2008). Novel Method of Doxorubicin-Spion Reversible Association for Magnetic Drug Targeting. International Journal of Pharmaceutics, 363(1-2), 170-176. https://doi.org/10.1016/j.ijpharm.2008.07.006
Nielsen, O. S., Horsman, M., and Overgaard, J. (2001). A Future for Hyperthermia in Cancer Treatment? European Journal of Cancer, 37(13), 1587-1589. https://doi.org/10.1016/S0959-8049(01)00193-9
Ortega, D., and Pankhurst, Q. A. (2013). Magnetic Hyperthermia. Nanoscience, 1, e88. https://doi.org/10.1039/9781849734844-00060
Panda, J., Satapathy, B. S., Majumder, S., Sarkar, R., Mukherjee, B., and Tudu, B. (2019). Engineered Polymeric Iron Oxide Nanoparticles as Potential Drug Carrier for Targeted Delivery of Docetaxel to Breast Cancer Cells. Journal of Magnetism and Magnetic Materials, 485, 165-173. https://doi.org/10.1016/j.jmmm.2019.04.058
Pankhurst, Q. A., Connolly, J., Jones, S. K., and Dobson, J. (2003). Applications of Magnetic Nanoparticles in Biomedicine. Journal of Physics D: Applied Physics, 36(13), R167. https://doi.org/10.1088/0022-3727/36/13/201
Pankhurst, Q. A., Connolly, J., Jones, S. K., and Dobson, J. (2003). Applications of Magnetic Nanoparticles in Biomedicine. Journal of Physics D: Applied Physics, 36(13), R167-R181. https://doi.org/10.1088/0022-3727/36/13/201
Pankhurst, Q., Thanh, N. T. K., Jones, S. K., and Dobson, J. (2009). Progress in Applications of Magnetic Nanoparticles in Biomedicine. Journal of Physics D: Applied Physics, 42(22), 224001. https://doi.org/10.1088/0022-3727/42/22/224001
Pardee, A. B., and Stein, G. S. (2009). The Biology and Treatment of Cancer: Understanding Cancer (300). John Wiley & Sons. https://doi.org/10.1002/9780470374252
Parsian, M., Unsoy, G., Mutlu, P., Yalcin, S., Tezcaner, A., and Gunduz, U. (2016). Loading of Gemcitabine on Chitosan Magnetic Nanoparticles Increases the Anti-Cancer Efficacy of the Drug. European Journal of Pharmacology, 784, 121-128. https://doi.org/10.1016/j.ejphar.2016.05.016
Perez, J. M., Josephson, L., O'Loughlin, T., Högemann, D., and Weissleder, R. (2002). Magnetic Relaxation Switches Capable of Sensing Molecular Interactions. Nature Biotechnology, 20(8), 816-820. https://doi.org/10.1038/nbt720
Perigo, E. A., Hemery, G., Sandre, O., Ortega, D., Garaio, E., Plazaola, F., and Teran, F. J. (2015). Fundamentals and Advances in Magnetic Hyperthermia. Applied Physics Reviews, 2(4), 041302. https://doi.org/10.1063/1.4935688
Perry, M. C. (2008). The Chemotherapy Source Book. Lippincott Williams & Wilkins.
Plank, C., Schillinger, U., Scherer, F., Bergemann, C., Rémy, J.-S., Krötz, F., Anton, M., Lausier, J., and Rosenecker, J. (2003). The Magnetofection Method: Using Magnetic Force to Enhance Gene Delivery. https://doi.org/10.1515/BC.2003.082
Portet, D., Denizot, B., Rump, E., Lejeune, J.-J., and Jallet, P. (2001). Nonpolymeric Coatings of Iron Oxide Colloids for Biological use as Magnetic Resonance Imaging Contrast Agents. Journal of Colloid and Interface Science, 238(1), 37-42. https://doi.org/10.1006/jcis.2001.7500
Pugazhendhi, A., Edison, T. N. J. I., Velmurugan, B. K., Jacob, J. A., and Karuppusamy, I. (2018). Toxicity of Doxorubicin (Dox) to Different Experimental Organ Systems. Life Sciences, 200, 26-30. https://doi.org/10.1016/j.lfs.2018.03.023
Rai, P., and Morris, S. A. (2019). Nanotheranostics for Cancer Applications. Springer. https://doi.org/10.1007/978-3-030-01775-0
Raj, K., and Moskowitz, R. (1990). Commercial Applications of Ferrofluids. Journal of Magnetism and Magnetic Materials, 85(1-3), 233-245. https://doi.org/10.1016/0304-8853(90)90058-X
Reddy, L. H., Arias, J. L., Nicolas, J., and Couvreur, P. (2012). Magnetic Nanoparticles: Design and Characterization, Toxicity and Biocompatibility, Pharmaceutical and Biomedical Applications. Chemical Reviews, 112(11), 5818-5878. https://doi.org/10.1021/cr300068p
Ren, F., Chen, J.-H., and Chen, Z.-L. (2005). Preparation and Characteristic Evaluation of Mitomycin C-Polybutylcyanoacrylate Magnetic Nanoparticles. Chinese Pharmaceutical Journal, 40(15), 1163.
Rocha-Santos, T. A. (2014). Sensors and Biosensors Based on Magnetic Nanoparticles. TrAC Trends in Analytical Chemistry, 62, 28-36. https://doi.org/10.1016/j.trac.2014.06.016
Ross, C. A. (2001). Patterned Magnetic Recording Media. Annual Review of Materials Research, 31(1), 203-235. https://doi.org/10.1146/annurev.matsci.31.1.203
Rudge, S., Kurtz, T., Vessely, C. R., Catterall, L., and Williamson, D. (2000). Preparation, Characterization, and Performance of Magnetic Iron-Carbon Composite Microparticles for Chemotherapy. Biomaterials, 21(14), 1411-1420. https://doi.org/10.1016/S0142-9612(00)00006-5
Rutnakornpituk, M., Baranauskas, V., Riffle, J., Connolly, J., St Pierre, T., and Dailey, J. (2002). Polysiloxane Fluid Dispersions of Cobalt Nanoparticles in Silica Spheres for use in Ophthalmic Applications. European Cells and Materials, 3(2), 102-105.
Salunkhe, A. B., Khot, V. M., and Pawar, S. H. (2014). Magnetic Hyperthermia with Magnetic Nanoparticles: A Status Review. Current Topics in Medicinal Chemistry, 14(5), 572-594. https://doi.org/10.2174/1568026614666140118203550
Savin, C.-L., Popa, M., Delaite, C., Costuleanu, M., Costin, D., and Peptu, C. A. (2019). Chitosan Grafted-Poly(Ethylene Glycol) Methacrylate Nanoparticles as Carrier for Controlled Release of Bevacizumab. Materials Science and Engineering: C, 98, 843-860. https://doi.org/10.1016/j.msec.2019.01.036
Scherer, F., Anton, M., Schillinger, U., Henke, J., Bergemann, C., Krüger, A., Gänsbacher, B., and Plank, C. (2002). Magnetofection: Enhancing and Targeting Gene Delivery by Magnetic Force in Vitro and in Vivo. Gene Therapy, 9(2), 102-109. https://doi.org/10.1038/sj.gt.3301624
Schultheiss-Grassi, P. P., Wessiken, R., and Dobson, J. (1999). TEM Investigations of Biogenic Magnetite Extracted from the Human Hippocampus. Biochimica et Biophysica Acta (BBA) - General Subjects, 1426(1), 212-216. https://doi.org/10.1016/S0304-4165(98)00160-3
Senyei, A., Widder, K., and Czerlinski, G. (1978). Magnetic Guidance of Drug-Carrying Microspheres. Journal of Applied Physics, 49(6), 3578-3583. https://doi.org/10.1063/1.325219
Shabani, Z., Morsali, A., Bozorgmehr, M. R., and Beyramabadi, S. A. (2019). Quantum Chemical Modeling of Iron Oxide Magnetic Nanoparticles Functionalized with Cytarabine. Chemical Physics Letters, 719, 12-21. https://doi.org/10.1016/j.cplett.2019.01.040
Shanmugavel, S., and Karthikeyan, V. (2014). Synthesis and Characterization of Layer by Layer Magnetic Nanoparticles of Methotrexate and Melphalan. World Journal of Pharmacy and Pharmaceutical Sciences, 3(2), 1809-1824.
Shen, T. T., Bogdanov, A., Bogdanova, A., Poss, K., Brady, T. J., and Weissleder, R. (1996). Magnetically Labeled Secretin Retains Receptor Affinity to Pancreas Acinar cells. Bioconjugate Chemistry, 7(3), 311-316. https://doi.org/10.1021/bc960003u
Simeonidis, K., Mourdikoudis, S., Kaprara, E., Mitrakas, M., and Polavarapu, L. (2015). Optimizing Magnetic Nanoparticles for Drinking Water Technology: The case of Cr(VI). Science of the Total Environment, 535, 61-68. https://doi.org/10.1016/j.scitotenv.2015.04.033
Song, H., Su, C., Cui, W., Zhu, B., Liu, L., Chen, Z., Zhao, G., Guo, Q., and Fan, L. (2019). Carboplatin Prodrug Conjugated Fe3o4 Nanoparticles for Magnetically Targeted Drug Delivery in Ovarian Cancer Cells. Journal of Materials Chemistry B, 7(3), 433-442. https://doi.org/10.1039/C8TB02574F
Song, L., Yang, K., Jiang, W., Du, P., Xing, J., Zhou, H., Ni, Y., and Li, X. (2020). Rituximab Conjugated Iron Oxide Nanoparticles for Targeted Imaging and Enhanced Treatment Against CD20-positive lymphoma. Journal of Materials Chemistry B, 8(5), 895-907. https://doi.org/10.1039/C9TB02521A
Sun, S., Murray, C. B., Weller, D., Folks, L., and Moser, A. (2000). Monodisperse FePt Nanoparticles and Ferromagnetic FePt Nanocrystal Superlattices. Science, 287(5460), 1989-1992. https://doi.org/10.1126/science.287.5460.1989
Tarantash, M., Nosrati, H., Kheiri Manjili, H., and Baradar Khoshfetrat, A. (2018). Preparation, characterization and in Vitro Anticancer Activity of Paclitaxel Conjugated Magnetic Nanoparticles. Drug Development and Industrial Pharmacy, 44(11), 1895-1903. https://doi.org/10.1080/03639045.2018.1508222
Tartaj, P., del Puerto Morales, M., Veintemillas-Verdaguer, S., González-Carreño, T., and Serna, C. J. (2003). The Preparation of Magnetic Nanoparticles for Applications in Biomedicine. Journal of Physics D: Applied Physics, 36(13), R182. https://doi.org/10.1088/0022-3727/36/13/202
Torchilin, V. P. (2000). Drug Targeting. European Journal of Pharmaceutical Sciences, 11, S81-S91. https://doi.org/10.1016/S0928-0987(00)00166-4
Toro-Cordova, A., Moreno-Vega, A. I., Araya-Durán, I., Araya, E., Chahua, M., Rinaldi, C., and Marican, A. (2018). Liposomes Loaded with Cisplatin and Magnetic Nanoparticles: Physicochemical Characterization, Pharmacokinetics, and in-vitro Efficacy. Molecules, 23(9), 2272. https://doi.org/10.3390/molecules23092272
Turner, R. D., Rand, R. W., Bentson, J. R., and Mosso, J. A. (1975). Ferromagnetic Silicone Necrosis of Hypernephromas by Selective Vascular Occlusion to the Tumor: A New Technique. The Journal of Urology, 113(4), 455-459. https://doi.org/10.1016/S0022-5347(17)59500-2
Ullah, M. F. (2008). Cancer Multidrug Resistance (MDR): A Major Impediment to Effective Chemotherapy. Asian Pacific Journal of Cancer Prevention, 9(1), 1-6.
Viota, J., et al. (2013). Functionalized Magnetic Nanoparticles as Vehicles for the Delivery of the Antitumor Drug Gemcitabine to Tumor Cells: Physicochemical in Vitro Evaluation. Materials Science and Engineering: C, 33(3), 1183-1192. https://doi.org/10.1016/j.msec.2012.12.009
Voit, W., Zapka, W., Belova, L., and Rao, K. (2003). Application of Inkjet Technology for the Deposition of Magnetic Nanoparticles to form Micron-Scale Structures. IEE Proceedings - Science, Measurement and Technology, 150(5), 252-256. https://doi.org/10.1049/ip-smt:20030692
Voltairas, P., Fotiadis, D., and Massalas, C. (2001). Elastic Stability of Silicone Ferrofluid Internal Tamponade (SFIT) in Retinal Detachment Surgery. Journal of agnetism and Magnetic Materials, 225(1-2), 248-255. https://doi.org/10.1016/S0304-8853(00)01265-8
Voltairas, P., Fotiadis, D., and Michalis, L. (2002). Hydrodynamics of Magnetic Drug Targeting. Journal of Biomechanics, 35(6), 813-821. https://doi.org/10.1016/S0021-9290(02)00034-9
Wang, J., Zhang, B., Yuan, Z., Han, S., Wang, C., and Chen, Y. (2011). Synthesis and Antitumor Efficacy of Daunorubicin-Loaded Magnetic Nanoparticles. International Journal of Nanomedicine, 6, 203. https://doi.org/10.2147/IJN.S16165
Weissleder, R., Bogdanov, A., Neuwelt, E. A., and Papisov, M. (1995). Long-Circulating Iron Oxides for MR Imaging. Advanced Drug Delivery Reviews, 16(2-3), 321-334. https://doi.org/10.1016/0169-409X(95)00033-4
Widder, K. J., Senyei, A. E., and Scarpelli, D. G. (1978). Magnetic microspheres: A Model System for Site Specific Drug Delivery in Vivo. Proceedings of the Society for Experimental Biology and Medicine, 158(2), 141-146. https://doi.org/10.3181/00379727-158-40158
Wu, M., Fan, Y., Lv, S., Xiao, B., Ye, M., and Zhu, X. (2016). Vincristine and Temozolomide Combined Chemotherapy for the Treatment of Glioma: A Comparison of Solid Lipid Nanoparticles and Nanostructured Lipid Carriers for Dual Drugs Delivery. Drug Delivery, 23(8), 2720-2725. https://doi.org/10.3109/10717544.2015.1058434
Xiong, H., Diao, Q., Jin, R., Song, D., Wang, X., and Sun, Y. (2017). Synthesis and Application of Thiol-Functionalized Magnetic Nanoparticles for Studying Interactions of Epirubicin Hydrochloride with Bovine Serum Albumin by Fluorescence Spectrometry. Luminescence, 32(2), 142-148. https://doi.org/10.1002/bio.3158
Xu, Y., et al. (2013). Bleomycin Loaded Magnetite Nanoparticles Functionalized by Polyacrylic Acid as a New Antitumoral Drug Delivery System. BioMed Research International, 2013. https://doi.org/10.1155/2013/462589
Yan, X.Q., Chen, J.H., Jiang, Q.F., Xi, N., Yang, L., nad Li, P. (2006). Preparation of Magnetic Dextran-Mitomycin Pro-Drug Nanoparticles. Chinese Journal of Hospital Pharmacy, 26(5), 507.
Yuan, W., Yuan, J., Zhou, L., Wu, S., and Hong, X. (2010). Fe3O4@poly(2-hydroxyethyl methacrylate)-graft-poly(ɛ-caprolactone) Magnetic Nanoparticles with Branched Brush Polymeric Shell. Polymer, 51(12), 2540-2547. https://doi.org/10.1016/j.polymer.2010.04.016
Zborowski, M., and Chalmers, J. J. (2011). Magnetic Cell Separation. Elsevier.
Zhang, Q., Wang, X., Cao, Y., Zhang, J., Guan, F., Chen, L., and Li, S. (2019). Cetuximab and Doxorubicin Loaded Dextran-Coated Fe3o4 Magnetic Nanoparticles as Novel Targeted Nanocarriers for Non-Small Cell Lung Cancer. Journal of Magnetism and Magnetic Materials, 481, 122-128. https://doi.org/10.1016/j.jmmm.2019.01.021
 This work is licensed under a: Creative Commons Attribution 4.0 International License
This work is licensed under a: Creative Commons Attribution 4.0 International License
© Granthaalayah 2014-2026. All Rights Reserved.